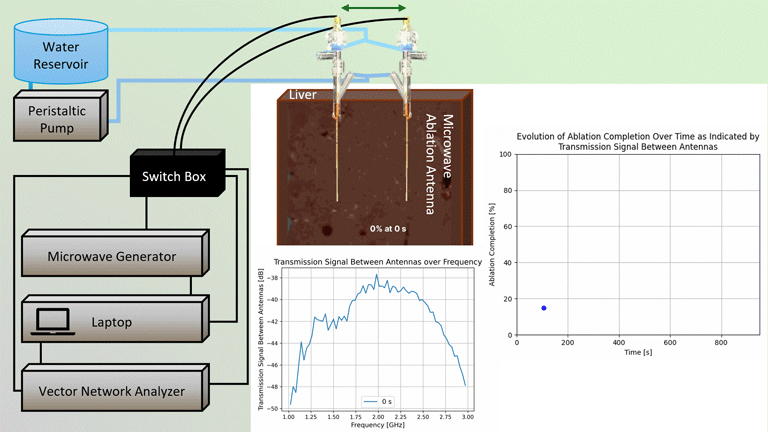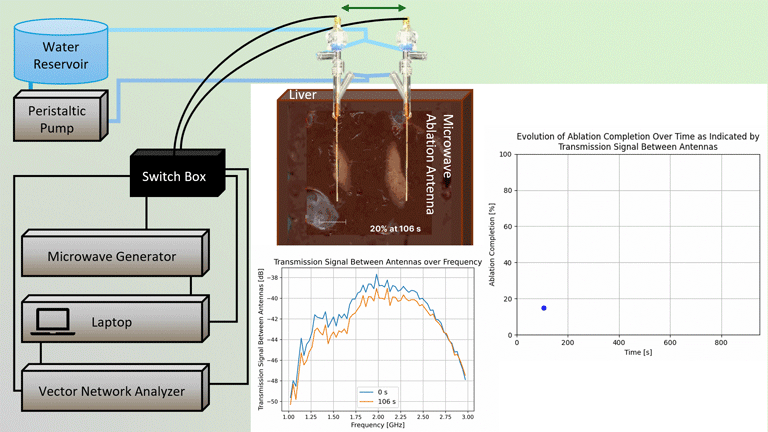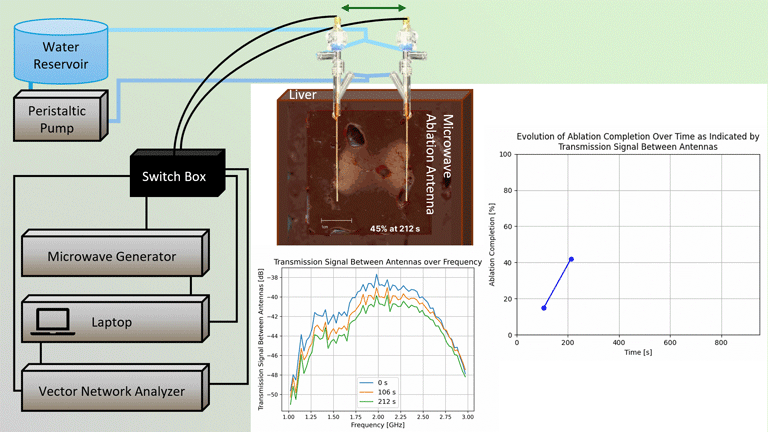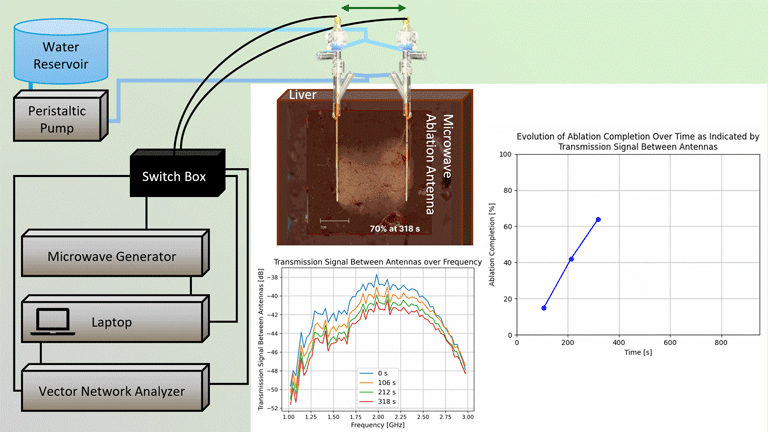
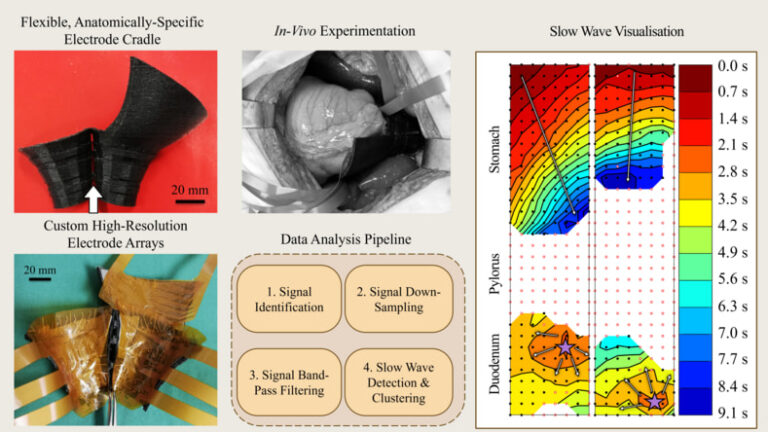
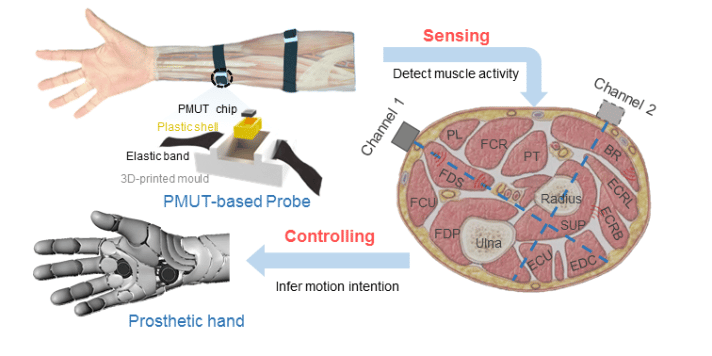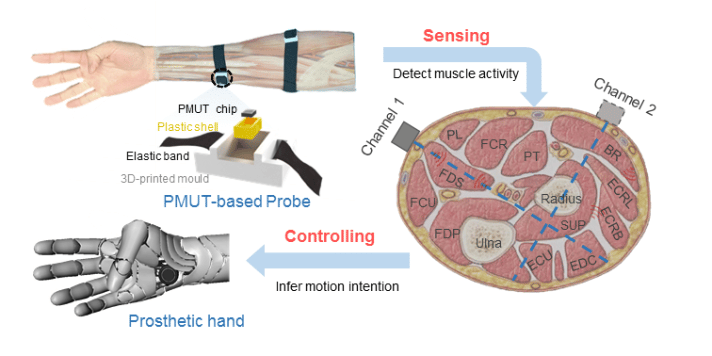
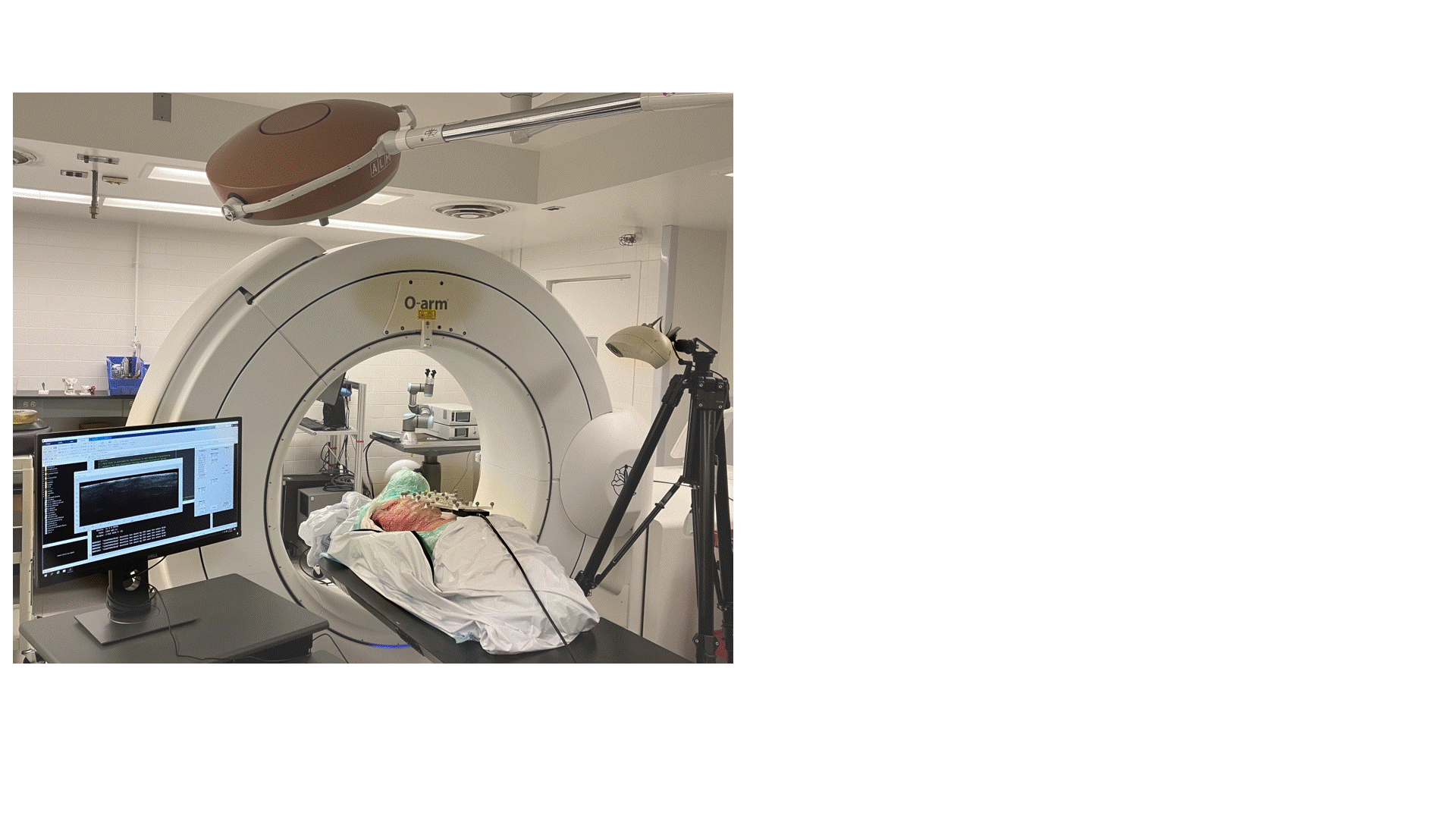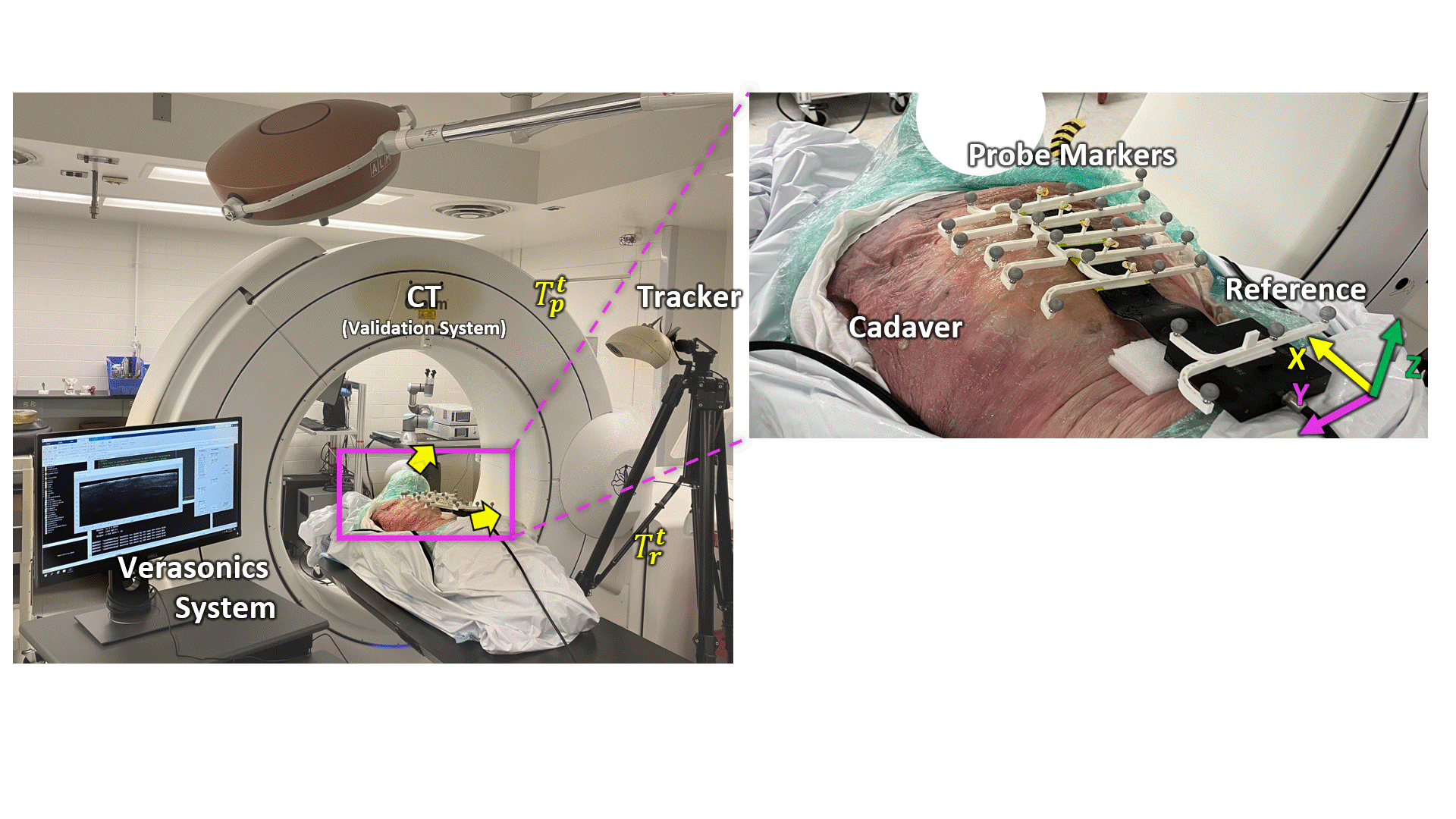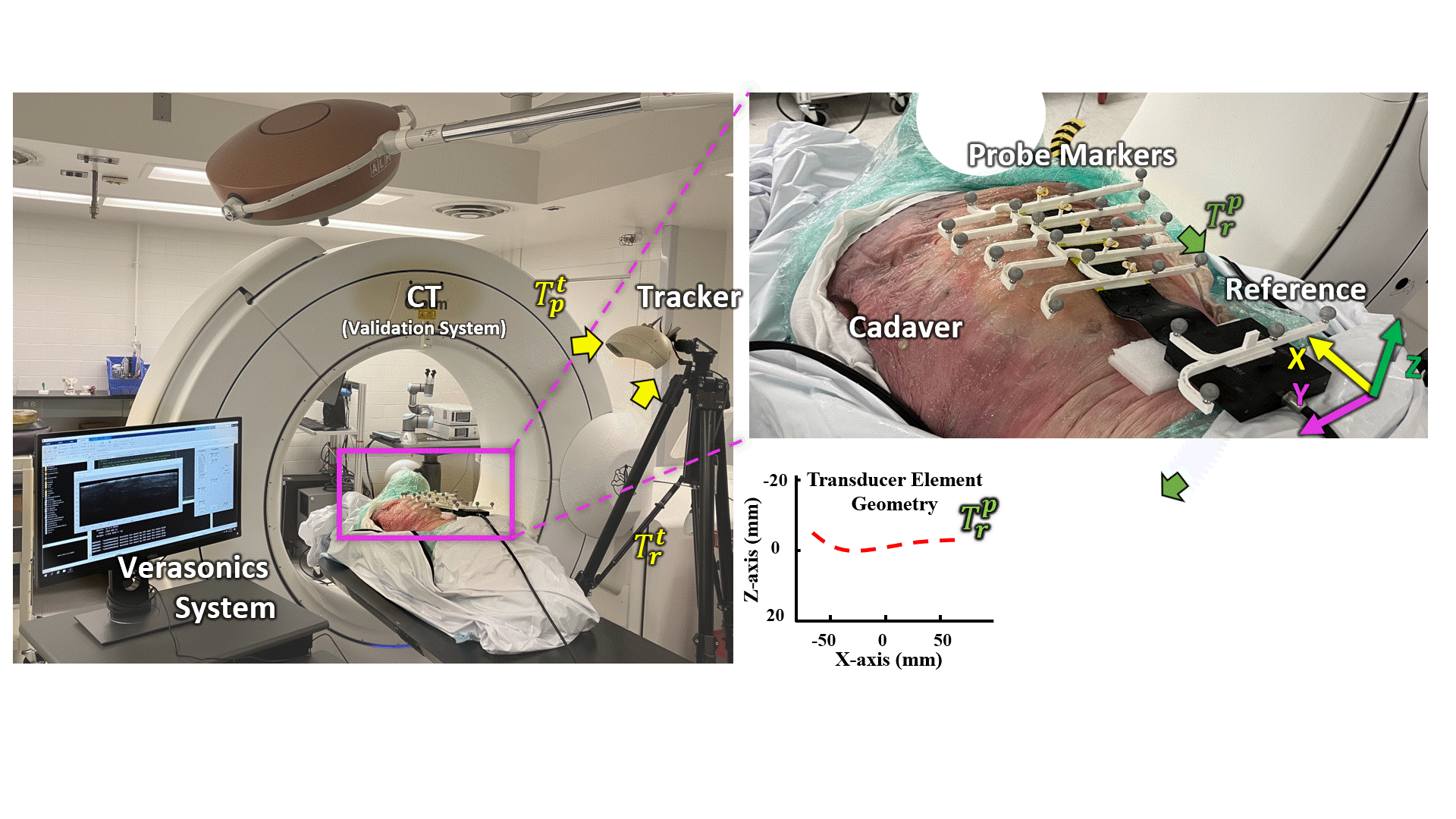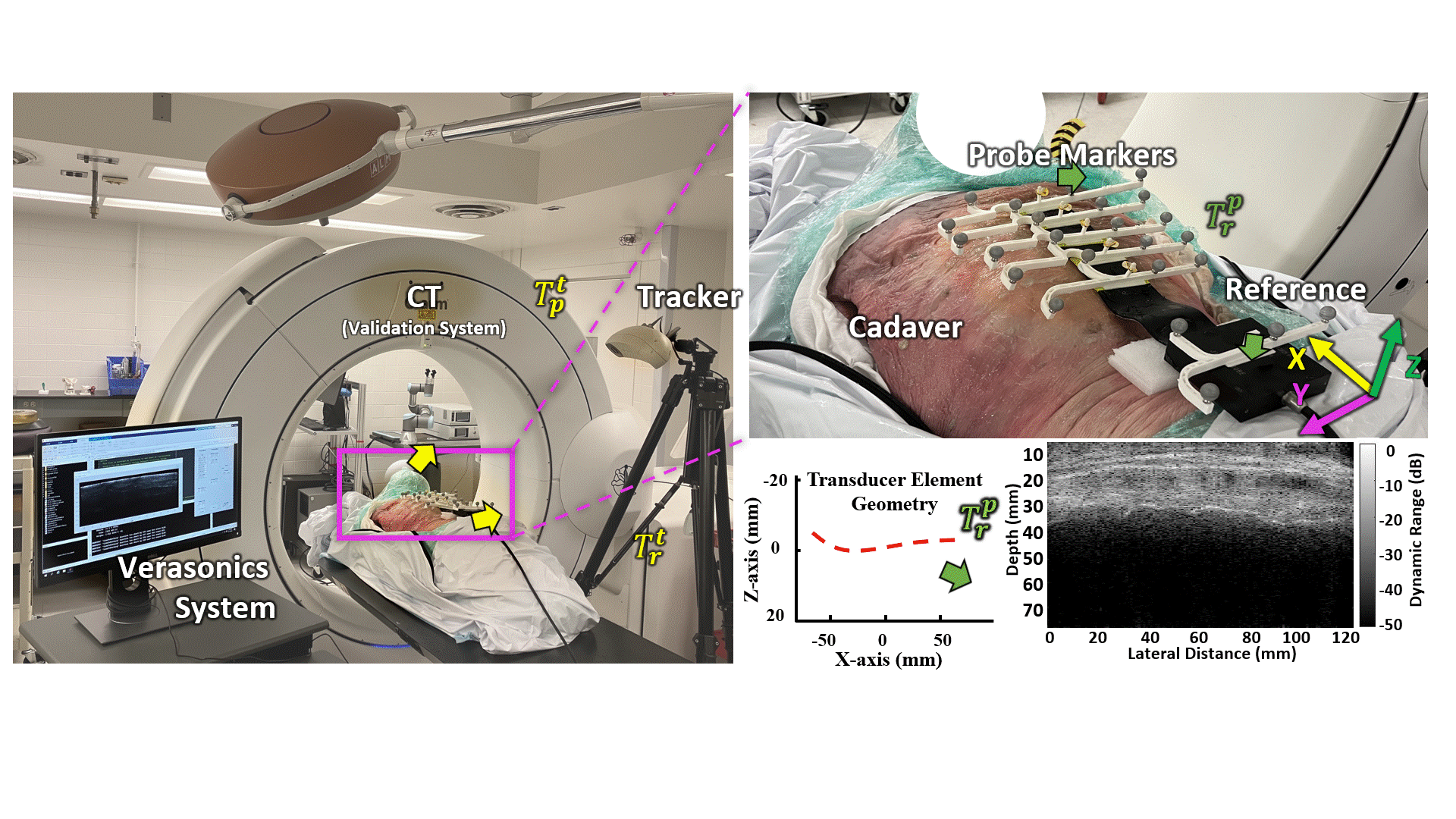
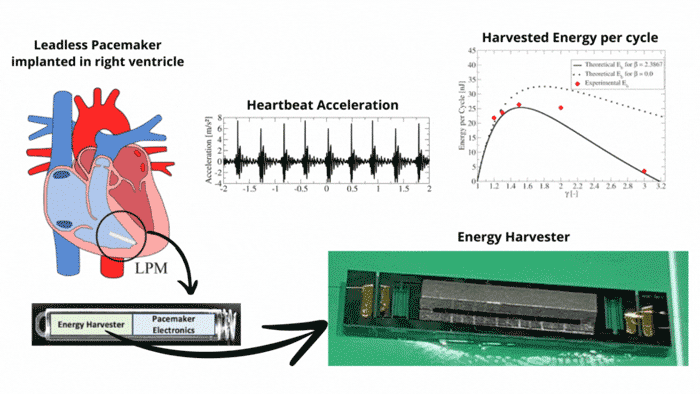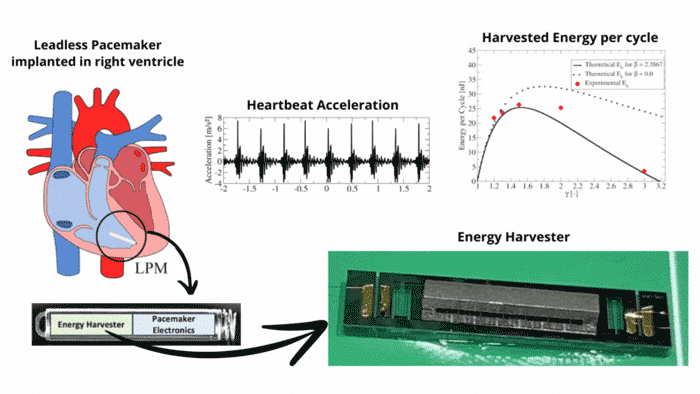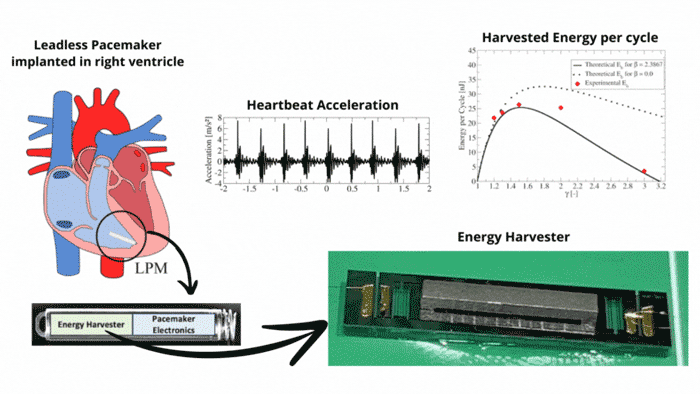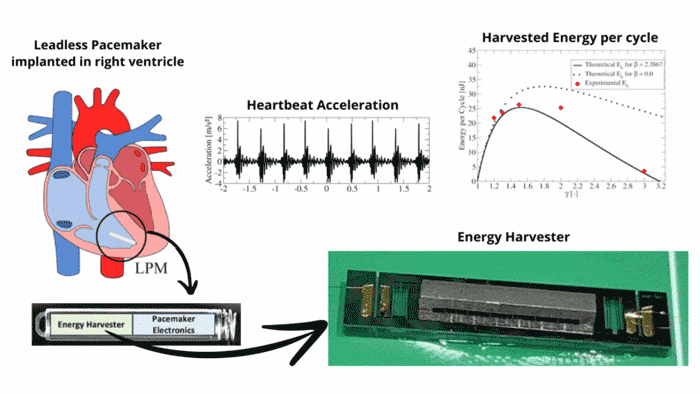
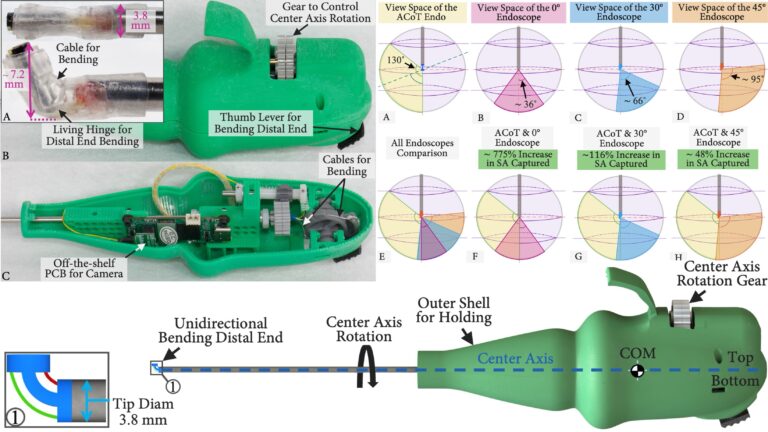
Transmission Coefficient-Based Monitoring of Microwave Ablation: Development and Experimental Evaluation in Ex Vivo Tissue
We demonstrate the feasibility of monitoring microwave ablation zone growth with the same antennas used for delivering ablation, potentially offering a means for real-time intraprocedural assessment.
read more